Researchers from the Center for Genomic Regulation in Barcelona and the University of Cologne have introduced an innovative experimental strategy to tackle fibrosis and scarring by obstructing collagen export at the cellular level. The team’s findings, published in the journal Nature Communications, demonstrate the strategy’s effectiveness, non-toxicity, and reversibility.
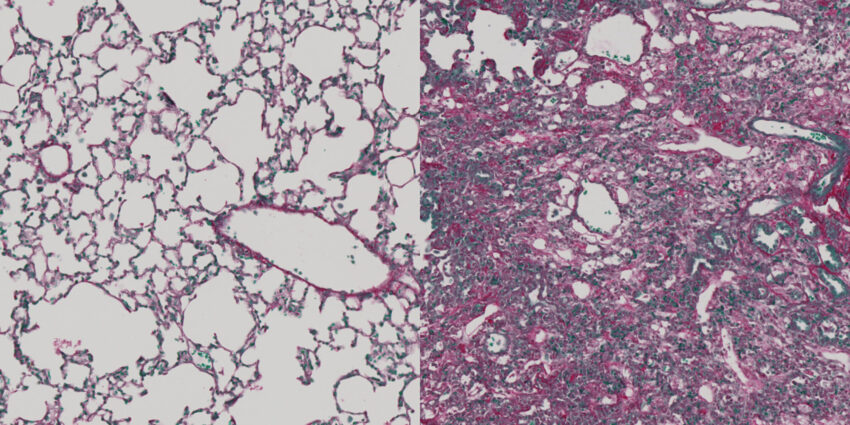
Scarring and fibrosis result from the secretion and accumulation of collagens between cells, typically as a response to injury or damage. Excessive collagen production can lead to fibrotic tissue, which can compromise the function of tissues and even entire organs. Approximately 45% of deaths in industrialized countries are linked to tissue fibrosis.
Current treatment options for scarring and fibrosis are limited to surgery. Topical creams are ineffective due to their inability to penetrate deeply enough to reach the affected areas and remodel the tissue. Inside the body, scarring and fibrosis can impact various tissues and organs, each with unique environments and challenges, making a one-size-fits-all treatment unfeasible.
The researchers’ novel approach focuses on using peptides to obstruct the export of collagen from within cells. They discovered that TANGO1 and cTAGE5, two proteins essential for collagen export, interact at the endoplasmic reticulum exit site. By targeting this site, the researchers aimed to achieve specificity and avoid the toxic effects associated with inhibiting the activity of the endoplasmic reticulum.
Proteins can be compared to puzzle pieces, and understanding how they connect at the molecular level is crucial for designing drugs that can block their interactions. However, the exact structure of TANGO1 and cTAGE5 remains unknown, making it challenging to develop drugs targeting their interaction.
To overcome this hurdle, the researchers employed AlphaFold2, an artificial intelligence program that can predict the shapes of proteins without requiring their 3D structural data. The AI-generated predictions enabled the researchers to design peptides capable of passing through cell membranes and disrupting the TANGO1-cTAGE5 interaction.
The team tested the peptides on normal human fibroblasts and observed a decrease in collagen export, with the effect being reversible within 48 hours. Similar results were obtained when testing fibroblasts from scleroderma patients and in zebrafish experiments.
The researchers plan to evaluate the efficacy of the peptides in pig skin and fine-tune their properties to enhance their potency. They believe this strategy could offer a new avenue for controlling collagen hypersecretion, potentially alleviating the cosmetic effects of skin scarring, treating autoimmune diseases like scleroderma, and manipulating post-surgery wound healing to prevent fibrosis.