Chemical Properties and Structure of Fumaric Acid
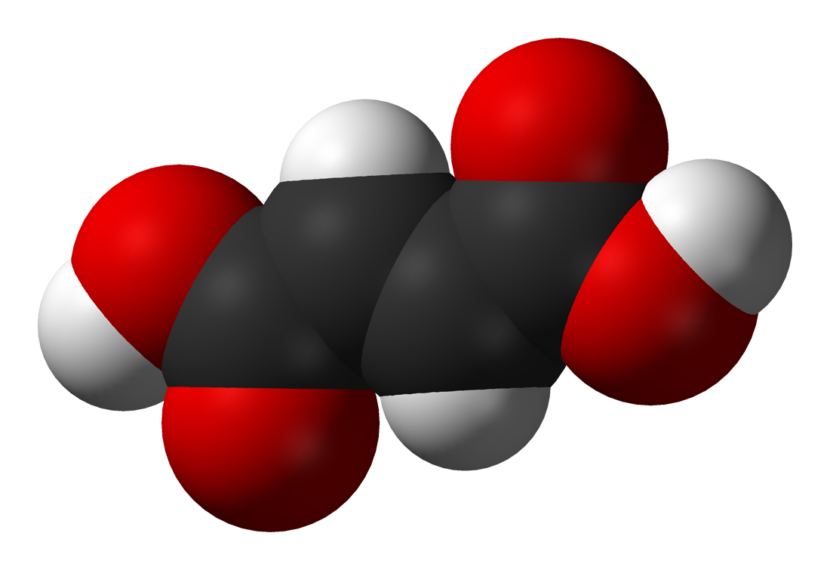
Fumaric acid is an organic compound that is best known by the chemical formula C4H4O4. It is a white crystalline solid that is relatively odorless and only slightly soluble in water. The structure of fumaric acid consists of a double bond between the two central carbons in the molecule, giving it a trans-butenedioic acid structure. This unique structure means fumaric acid exists as two geometric isomers – trans-fumaric acid and cis-fumaric acid. However, the trans isomer is the only naturally occurring isomer and is about 1000 times more stable than the cis isomer.
Natural Sources and Production of Fumaric Acid
Naturally, Fumaric Acid can be found in certain plants, such as lichens and fumitory plants. It is an intermediate product in the Krebs citric acid cycle, which is important in cellular respiration. On an industrial scale, fumaric acid is produced synthetically through a hydration reaction between maleic anhydride and water. Maleic anhydride is derived from benzene or butane and oxidized to form the anhydride intermediate. Nearly 90% of global fumaric acid supply is produced via this maleic anhydride route. Smaller amounts are also obtained through fermentation of carbohydrates by certain fungi. China and Germany are currently among the top producers and suppliers of fumaric acid worldwide.
Applications in Food Additives and Supplements
One of the biggest uses of fumaric acid is as a food additive. As it has acidic properties but is considered safe for human consumption, it serves as an effective pH regulator and preservative in various food and beverage products. Fumaric acid, or its calcium, potassium, or sodium salts, are commonly added to processed meats, baked goods, condiments, and drinks. It helps extend shelf-life by inhibiting microbial growth. Fumaric acid supplements are also popular as they may promote feelings of fullness when consumed and help control appetite. Some research has indicated it could help in weight management when taken as part of a calorie-controlled diet.
Roles in Animal Feed and Skin Care
Due to its antibacterial properties, fumaric acid finds widespread application as an additive in animal feed production. It acts as a preservative and helps ensure feed remains fresh for longer periods during storage and transport. Fumaric acid is approved for direct addition to cattle, poultry, pig, and fish feeds. In skin care products, fumaric acid functions as an exfoliant due to its keratolytic properties. It helps remove dead skin cells from the surface while also reducing inflammation caused by psoriasis and other skin conditions. Many creams, serums, and soaps contain fumaric acid or its esters for exfoliating and moisturizing benefits.
Use as a Chemical Intermediate
Apart from direct applications, fumaric acid serves as an important chemical intermediate in various industrial processes. It undergoes esterification reactions to produce monoethyl fumarate and dimethyl fumarate, which are common plasticizers and solvents. Upon heating, fumaric acid forms maleic anhydride through dehydration, providing an economical route for manufacturing this versatile anhydride. Fumaric acid also acts as a precursor in producing important compounds like aspirin, glycerides, and alkyd resins. The double bond in its structure allows it to participate in both addition and condensation polymerization reactions, generating a class of thermosetting resins known as fumaric acid resins.
Medical Significance of Fumaric Acid Esters
Recent research has revealed fumaric acid esters have anti-inflammatory and immunomodulating properties, making them useful therapeutic agents. Dimethyl fumarate, in particular, shows considerable potential. It is believed to stimulate antioxidant and anti-oxidative stress mechanisms within cells. Studies indicate it can effectively treat the skin condition psoriasis and moderate cases of multiple sclerosis. With fewer side effects than alternatives, dimethyl fumarate has gained approval as an oral medicine for relapsing-remitting multiple sclerosis in many countries under the trade name Tecfidera. Investigations are ongoing into whether it may assist in managing other autoimmune or neurodegenerative illnesses too.
Safety Considerations for Industrial Applications
While generally recognized as safe to ingest, fumaric acid requires prudent handling as its concentrated dry form or dust can cause minor skin, eye, and respiratory irritations upon direct contact. Workers dealing with bulk quantities are advised to wear proper protective equipment like goggles, gloves, and respirator masks. Its solutions also tend to be corrosive to certain metals and degrade or dissolve many plastics over time. Close monitoring and corrosion-resistant containers are needed when fumaric acid is stored or transported. All disposal and discharge should follow environmental regulations to prevent aquatic toxicities. With care taken in manufacturing and usage, the versatile chemical continues making vital contributions across sectors.
In conclusion, this article has discussed the chemical and physical properties of fumaric acid, outlined its industrial synthesis methods and natural sources, and explored its wide applications pertaining to food, animal feed, skin care, chemical intermediacy, and medical significance. Whether serving as a preservative, plasticizer, or therapeutic agent, fumaric acid’s unique double bond structure and reactivity profile lend it indispensable functionality. Its versatility continues enabling progress.